
CLASSIFICATION OF MAGNETIC SUBSTANCES
Date: 2015-10-07; view: 766.
Substances can be classified as belonging to one of three categories, depending on their magnetic properties. Paramagneticand ferromagneticmaterials are those made of atoms that have permanent magnetic moments. Diamagneticmaterials are those made of atoms that do not have permanent magnetic moments.
Ferromagnetism
A small number of crystalline substances exhibit strong magnetic effects called ferromagnetism.Some examples of ferromagnetic substances are iron, cobalt, nickel, gadolinium, and dysprosium. These substances contain permanent atomic magnetic moments that tend to align parallel to each other even in a weak external magnetic field. Once the moments are aligned, the substance remains magnetized after the external field is removed. This permanent alignment is due to a strong coupling between neighboring moments, a coupling that can be understood only in quantum-mechanical terms.
All ferromagnetic materials are made up of microscopic regions called domains,regions within which all magnetic moments are aligned. These domains have volumes of about 10-12to 10-8m3 and contain 1017 to 1021atoms. The boundaries between the various domains having different orientations are called domain walls.In an unmagnetized sample, the magnetic moments in the domains are randomly oriented so that the net magnetic moment is zero. When the sample isplaced in an external magnetic field 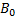 , the size of those domains with magnetic moments aligned with the field grows, which results in a magnetized sample. As the external field becomes very strong, the domains in which the magnetic moments are not aligned with the field become very small. When the external field is removed, the sample may retain a net magnetization in the direction of the original field.
, the size of those domains with magnetic moments aligned with the field grows, which results in a magnetized sample. As the external field becomes very strong, the domains in which the magnetic moments are not aligned with the field become very small. When the external field is removed, the sample may retain a net magnetization in the direction of the original field.
At ordinary temperatures, thermal agitation is not sufficient to disrupt this preferred orientation of magnetic moments. When the temperature of a ferromagnetic substance reaches or exceeds a critical temperature called the Curie temperature,the substance loses its residual magnetization and becomes paramagnetic. Below the Curie temperature, the magnetic moments are aligned and the substance is ferromagnetic. Above the Curie temperature, the thermal agitation is great enough to cause a random orientation of the moments, and the substance becomes paramagnetic.
| <== previous lecture | | | next lecture ==> |
| Reading and Writing | | | Paramagnetism |